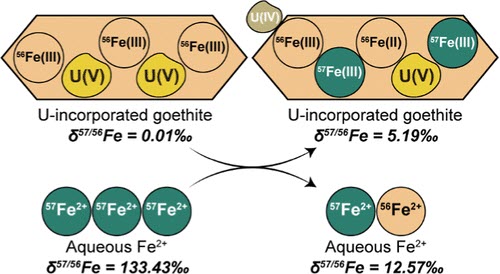
Iron (oxyhydr)oxides are capable of structurally incorporating and releasing trace elements through Fe2+-induced recrystallization. However, estimation of the extent of recrystallization varies among studies using isotopic tracers and those examining the release of occluded trace elements. To provide further insight into the dynamics of Fe2+-catalyzed iron (oxyhydr)oxide recrystallization, and to accurately interpret the isotopic and elemental compositions of iron (oxyhydr)oxides in the rock record, we probed the coupled dynamics of goethite isotopic exchange and trace element cycling during Fe2+-induced recrystallization. Herein, 57Fe labeled Fe2+ was reacted with U(V)-incorporated goethite, and the resulting isotopic and elemental interactions were systematically characterized and modeled. Partial release of incorporated U (3.9%) was observed, which suggested limited recrystallization while Fe isotope measurements suggested nearly complete isotopic mixing after 30 days. A box model incorporating partial and gradual back-reaction estimated an overall goethite recrystallization extent of ~32.8%, narrowing the gap to the observed U release (3.9%) as compared to the traditional fast back-reaction-only model (88%). This study highlights the role of Fe2+-driven recrystallization in controlling trace metal cycling and provides insights into the stability and reactivity of U-incorporated goethite in reducing environments. The findings refine our understanding of iron (oxyhydr)oxide recrystallization processes and their implications for trace element mobility in natural systems.
Article link: https://doi.org/10.1021/acs.est.5c11748





 Address
Address
 E-Mail
E-Mail
 Telephone
Telephone